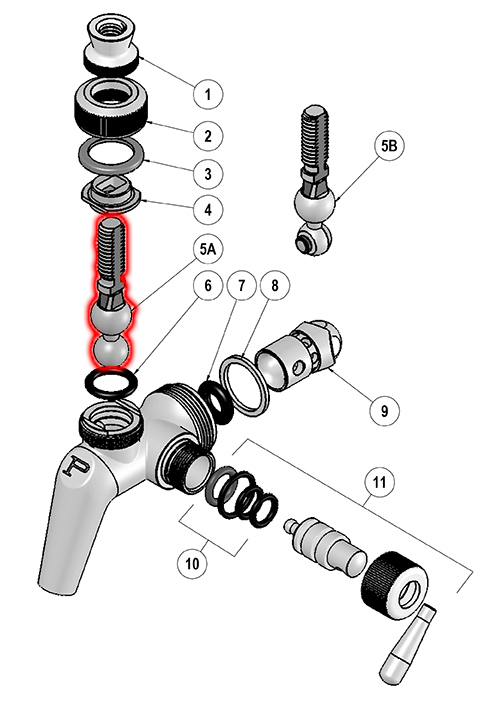
I had some time over the weekend to experiment further in order to identify which component of the faucet was at fault, and whether the hypothesis that acidity being the catalyst was correct. I placed each separate piece of the faucet into its own container filled with a StarSan solution measured via a calibrated Omega PHH-7011 to a pH of 2.45. After 5 minutes I removed each piece and smelled them individually to try and detect if any emitted a rotten egg smell. All parts except one failed to emit the odor. Item 5A, Part No. 68726, "Handle Leaver" is the culprit; shown in red in the following image, it reeks of rotten eggs. Subsequent tests via dipping in StarSan caused the aroma of rotting eggs to become present in as little as 10 seconds.
In order to test the acidity hypothesis I prepared three solutions: A sample of tap water treated with Phosphoric acid to a pH of 2.51, and sample of tap water treated with Lactic Acid to a pH of 2.57, and a sample of ordinary bottled Coke, with a pH of 2.37. Having three 650SS faucets, I placed one of the Handle Leaver's of each faucet into each of the three solutions, leaving them to soak for around 4 hours. So far the Coke and Phosphoric solution emit
no noticeable aroma. The Lactic acid sample does smell faintly sulphuric. This evidence suggest that the hypothesis that all acidic solutions produce the rotten egg aroma is actually
false. But that some active ingredient in StarSan (possible the surfactant Dodecylbenzenesulfonic Acid), or Lactic Acid (a notable factor in many sour beers) is still reacting with the Handle Leaver's in these taps, causing the rotten egg smell. I will continue to monitor these samples and report back if any of them change.
Still waiting on word back from Perlick. I have directed them to this post, as well as the other posts previously linked.





