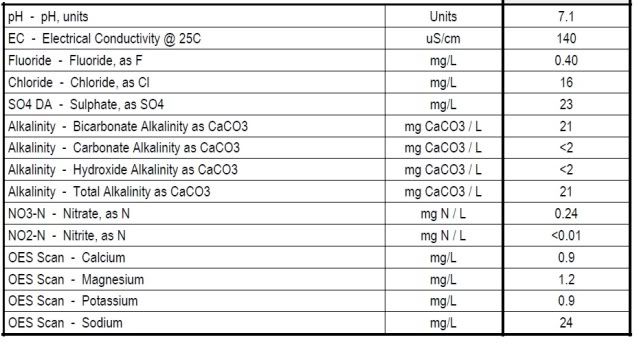
Main reason as far as I understand is that salts are used to alter the mash pH and in the boil will affect yeast health and final flavour.
Some people disagree but I am generally not a fan of carbonates, especially not in beers that aren't black* (reading supports this). Thinking about it logically, the pH is dropped by the calc chloride and calc sulphate and naturally dropped and buffered by the mash. Calc carbonate raises the pH and works against that (and its flavour contribution isn't great either.
Work for a mash pH of between 5.2 and 5.4 using just the calc chloride and sulphate and see how far away from your intended profile.
Personally I'd just work with those two to get the malt/hop profile you want and forget about a possibly dodgy, incorrect or redundant water profile. Some people suggest sulphates don't work well with noble hops. I haven't noticed this but need to try a side by side. You could then use an acid addition (lactic, citric, phosphoric etc) or a small amount of acidulated malt to get the pH down along with the calc chloride.
* I don't put them in black beers either.